
Sourdough Fermentation Process – How Does It All Work?

I’ve heard top, top bakers, some of the world’s most celebrated, talk absolute nonsense about the sourdough fermentation process. So I feel it’s my duty to provide clarity on this topic. To be fair, it’s easy to be confused about how sourdough fermentation works. There are so many processes going on. And even in this fairly hefty article, I haven’t delved into the detail of them all. One thing is for sure is that it’s not just yeast that makes sourdough rise. Sourdough is much more complicated!
In this article, I aim to accurately explain the science behind the sourdough fermentation process without being too technical. I mean, the subject is simple stuff for biologists, and the average home baker could spend a fortnight collecting data on it, so I’ve done it for you!
Before we start, this is not a guide on best practices or sourdough troubleshooting, and it won’t answer questions such as, “why is my sourdough bread dense?” – directly anyway. The purpose of this guide is for bakers to understand what is really going on when making sourdough bread. With this knowledge, you’ll make better choices to improve your bread. So let’s get into how sourdough works by understanding the basics first.

What is sourdough?
A levain (or “leaven” depending on what country you’re from) is the active ingredient used in a bakery product to make it rise. In the case of making bread, the levain produces carbon dioxide, which is then trapped by interlocking gluten strands. As more CO2 is produced, the pockets of gluten expand to make the dough rise.
In the past couple of centuries, bakers have changed from using natural sourdough levain to baker’s yeast. The particular yeast strain, saccharomyces cerevisiae, is used in all varieties of bread baking yeast. But as I’m sure you’re aware, the use of sourdough has been revitalised in recent years. Though not nearly as widespread as yeasted bread, sourdough is extremely popular to make at home, and it’s a popular item across the world.
Sourdough bread takes longer to rise than yeast-leavened bread. This (in part) provides sourdough bread with its unique flavour, texture and health benefits, which we’ll discuss in this article.
There are lots of supermarket imitation sourdough bread. The UK’s real bread campaign regularly tarnishes big brands who attempt to pass yeast bread off as sourdough. In many cases, baker’s yeast and dough conditioners are used alongside sourdough, which adds dough maturity for rapidly produced bread.
Sourdough bread doesn’t require extra ingredient additions to make it perfect. Aside from a little malt flour in some cases, all that is required to make outstanding sourdough bread is flour, water, salt and a starter made from fermenting flour and water. An experienced baker can manipulate their techniques to produce unique flavours and extraordinary bread. And, after reading this, you’ll be able to do the same.
What is in a sourdough starter?

A sourdough starter is, in essence, a culture of wild yeasts and lactic bacteria. You can learn how to make a sourdough starter on my recipe page, but I’ll skim over the basics.
Water and flour are combined in a clean jar and left for 24 hours. The next day, ⅔’s of the mixture is replaced with fresh flour and water. The discarding and refreshment steps repeat every 24 hours or when it peaks (rises to the top).
Over time, wild yeast and bacteria become more and more active. This can be witnessed by the starter rising as it produces gas. After a couple of weeks, a mature starter that’s ready to make bread is produced.
How does sourdough produce gas?
The wild yeasts and Lactic Acid Bacteria (LAB) found in a starter get introduced primarily in the flour. Yet, some of the microflora are airborne. Scientists dispute the effects of flour vs environment vs bacteria from bakers’ hands. But the most trusted sources (in my opinion) say the selection of bacteria and wild yeasts in a starter is largely contributed by the flour used.
Over time, the number of yeasts and LAB in a new starter increases. They produce enzymes to break down carbohydrates in the flour to create simpler sugars. The yeast and LAB process these small sugars to produce gas and other beneficial products for the dough.
Each starter will have a unique blend of different species and strains of yeast and lactic bacteria. Each one will vary in activity, and as the starter matures, the species will change. Those better suited to the progressively acidic environment become more prominent. This is most apparent in the first week of a starter, where its activity levels fluctuate as the species of bacteria and yeast compete with each other.
In most mature starters, LAB outnumbers yeast cells by 100 to 1. However, carbon dioxide is produced by LAB at a slower rate when compared to yeast. This means that LAB and wild yeasts are both responsible for gas production at (approximately) a 50:50 ratio.
What is Lactic Acid Bacteria?
Lactic Acid Bacteria (LAB) are rod-shaped (bacilli) or spherical (cocci) bacteria found in many living things, primarily decomposing plants and milk products. The main species in sourdough is Lactobacillus, but many other lactic acid bacteria genera exist.
They provide enzymes and catalysts for the sourdough fermentation process to produce lactate alongside other outputs. Lactate creates lactic acid when it takes a hydrogen ion. Fortunately, hydrogen is readily available in bread dough. Because of how easy lactate can produce lactic acid, bread bakers often use each term interchangeably.
Wild yeasts and LAB are aerotolerant anaerobes. Meaning they organically multiply in the presence of oxygen. Airflow is essential to a new starter, created by loosening the lid of the jar. Another reason for a loose-fitting cover is the rising air pressure from the CO2 released, which can force a glass jar to explode! For this second reason, only loosely seal the lid of a mature starter too.
NOTE: Lactic acid bacteria will also appear in long-fermented yeast-leavened bread. Preferments such as biga or poolish use the organic acids produced to enhance the maturity of the bread.
Stage 1: Breaking down starch into sugars
To produce gas, yeast and organic acids need a carbon source. For this, we need to turn the carbohydrates in the flour into simpler sugars.
Starch is the carbohydrate that makes up around 70-75% of the flour. For the starch to be metabolised by yeast and bacteria, it must be broken down. Starches are technically polysaccharides. These are chains of bonded monosaccharides, the scientific name for single sugars.
To produce sugar for gas production, polysaccharides get broken into monosaccharides (single sugars) or disaccharides (two single sugars bonded).
Different sugars are produced depending on the yeast species and LAB in the starter. The 6-cell carbon sugar group, hexoses, are the most widely found monosaccharide type in bread and includes glucose and fructose sugars. The following section explains how starches are broken down.

Amylase
The amylase enzyme is the kickstarter to break down starch and already exists in the flour. If low volumes of amylase are in the flour, adding malt flour or fungal amylase provides a similar diastatic boost. Maybe you have noticed these ingredients on flour packaging?
Amylase gets to work immediately by breaking down the damaged starch in the flour. In most flours, 10-15% of starch is damaged, so this is no problem! There are two versions of amylase; ɑ-amylase and β-amylase:
ɑ-amylase
ɑ-amylase takes substrate polysaccharides, namely, amylose (22-25% of the starch) and amylopectin (75-78%), and breaks them down into maltose, maltotriose (chains of the disaccharide, maltose), glucose and dextrins. Its peak output occurs between 5-6 pH. Therefore, as a starter ages and becomes more acidic, ɑ-amylase productivity slows.

β-amylase
β-amylase has an important role, too. It:
- Breaks the maltotriose chains to produce maltose
- Picks off maltose to be consumed by the yeast or LAB
- Breaks down dextrins into glucose
Maltose
Many yeast and LAB produce maltase, which is an enzyme that breaks up the maltose disaccharide into two glucose molecules.
As with all of these processes, the simple sugars created are dependent on how certain starches are broken down. I will share an example later regarding the popular bacteria lactobacillus sanfranciscensis.
Dextrins
Dextrins are great! These starches soak up water meaning extra water is added to the recipe when flour contains a high quantity of dextrins. They make production costs cheaper, a moister crumb and a more-even oven spring. It’s not a surprise that extra dextrin is commonly added to commercial bread recipes!
Dextrins can be broken down into glucose by β-amylase. But, when acidic sourdough bread is in the oven, any dextrins that have not degraded become hydrolysed. In hydrolysis, the short-chained starch partially rebranch with the degraded starch molecules in the flour. This leads to the starch hardening and darkening as part of the Maillard reaction. This process contributes to the flavour, colour and crispness of the crust.
Glucofructan
Flour’s most prevalent starches are glucofructans, which contribute to 1-2% of the dough. They consist of one fructose molecule chained to at least one glucose molecule. Yeast will produce enzymes such as invertase and sucrase to remove the glucose molecules from the chain. Eventually, all the glucose and fructose molecules are separated and available for processing by the levain.
Sucrose (table sugar) is the simplest type of glucofructan. It consists of a bond of just one glucose molecule to one fructose. Enzymes, invertase and sucrase break down bonds to separate glucose and fructose molecules into individual components.
Step 2: Glycolysis breaks down sugars for respiration and fermentation
The enzymes mentioned above describe pathways to produce the simple sugars, glucose and fructose. After separation, the sugars enter a metabolic pathway called glycolysis. The first section of this is the Embden-Meyerhof Pathway (EMP).
You will notice ATP and ADP in the diagram. Adenosine Triphosphate (ATP) is the energy-carrying molecule in all living cells. It donates molecules to other cells for reactions to happen. In bread baking, ATP releases phosphate molecules to create Adenosine Diphosphate (ADP), a less prolific energy-carrying molecule.
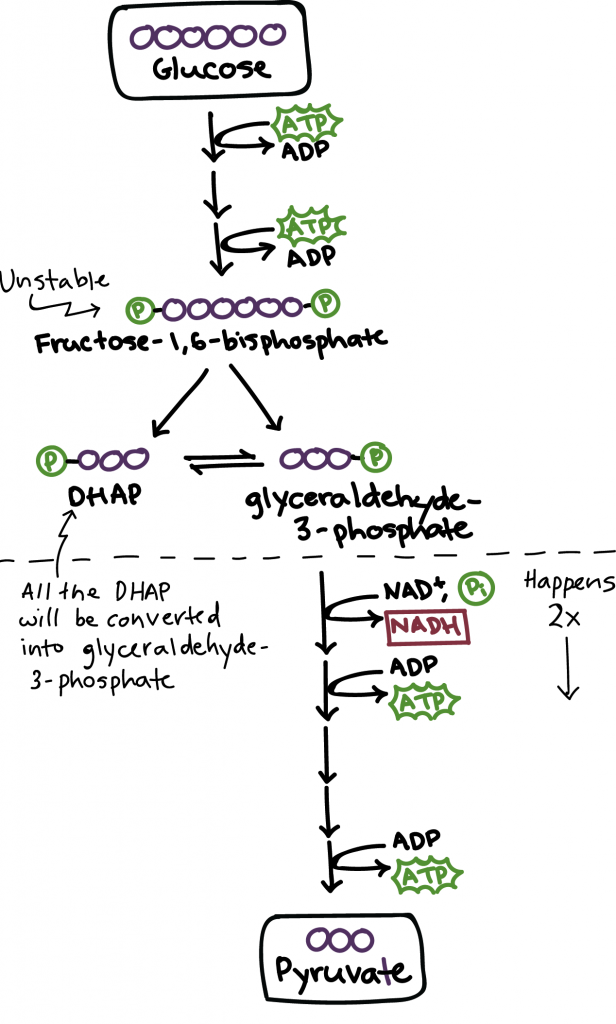
There are two stages of glycolysis. In the energy-requiring phase:
- The glucose molecule is rearranged.
- Two phosphate molecules latch onto the glucose.
- This transforms the molecule into fructose-1,6-bisphosphate.
- Phosphates are taken from two ATP energy molecules.
- ATP is then converted into the less powerful energy molecule, ADP.
- The modified sugar is now unstable and thus splits into two phosphate-bearing three-carbon sugars.
- One sugar is DHAP, and the other is glyceraldehyde-3-phosphate.
- Only glyceraldehyde-3-phosphate can pass onto the next stage of glycolysis.
- The DHAP sugar is easily converted into glyceraldehyde-3-phosphate, making the output of EMP, 2 glyceraldehyde-3-phosphates.
The second stage of glycolysis is the Energy-Releasing-Phase. This cycle occurs twice as EMP produced two three-carbon Glyceraldehyde-3-phosphate sugars. Both sugars undergo a series of reactions catalysed by several enzymes that utilise NAD+, a coenzyme found in all living cells that is vital for metabolism.
NAD+’s main function in glycolysis is to transfer hydrogen back and forth. When NAD+ is reduced (by gaining a hydrogen molecule), it becomes NADH. When NADH passes on the hydrogen, it reverts to NAD+.
Several reactions affect the sugars, including reinstating the phosphate molecules to ADP, which produces two more of those energetic ATP molecules. In doing so, water (H2O) is excreted, and two pyruvates remain.
In our bodies, ATP is what gives us the energy to move. But the pyruvate is the key component for the next stage of bread making. By the end of the process, we are left with two pyruvates, 2 ATP, 2 H2O and 2 NADH. The full equation is displayed here:
C6H12O6 + 2 ADP + 2 Pi + 2 NAD+ → 2 CH3COCOO− + 2 ATP + 2 NADH + 2 H2O + 2 H+
*Glucose is C6H12O6 & Pyruvate is CH3COCOO−
Providing there is a continual serving of simple sugars, glycolysis is repeated. Other monosaccharides that are produced by enzymes, such as fructose, ribose, gluconate and (possibly) arabinose can also follow glycolysis.
To make dough rise, the pyruvate needs to produce carbon dioxide. There are several ways to do this in bread dough:
Respiration
In the presence of oxygen, yeast is able to respire. In this route, pyruvates follow the Krebs or Citric-acid cycle to release carbon dioxide. It is a complicated chain of reactions where pyruvate is decarboxylated to make acetyl. The acetyl is oxidised and undergoes a cycle of reactions that release CO2. At the end of the process, energy in the form of ATP is produced, as well as more acetyl. If oxygen is still present, the produced acetyl is re-oxidised, and the cycle repeats.

Note: Carbon dioxide is transported as a liquid to a weak point in the gluten before being released as gas. The gluten matrix captures the gas, and the dough expands, making it rise.
Step 3: Fermentation processes
Respiration is the fastest way to raise bread as it produces energy and CO2 in high volumes. Compared to yeast, sourdough takes longer to rise. As dough rises, it continues to absorb a small amount of oxygen. Oxygen is vital for yeast respiration, but too much will destroy flavour and weaken the dough structure, which occurs when dough is over-oxidated.
Kneading is the most significant contributor to oxidation. When making bread commercially, high-intensity mixers and oxidating additives (ascorbic acid or propane bromate) increase oxygen intake. When making sourdough bread, gentle kneading or no-knead methods are used to reduce the amount of oxygen in the dough. Less oxidation means that the first rise can be extended without the risk of the dough weakening. However, the lack of oxygen means the yeast cannot respire.
Without oxygen, pyruvates cannot follow the Krebs cycle, so must take a different path. Anaerobic respiration leads to fermentation. In sourdough, this will be initiated by the yeast for alcoholic fermentation or by lactic bacteria utilising homofermentative or heterofermentative pathways. Some routes produce CO2, but not all. Yet, they are vital for making terrific sourdough bread!
Alcoholic fermentation
As discussed in the bread fermentation process article, respiration and alcoholic fermentation produce gas in yeasted bread. Alcoholic fermentation is only a handful of steps away from respiration. It happens when no oxygen is available to conduct the Krebs cycle after EMP.

The pyruvate produced in glycolysis latches to an inorganic phosphate. At the same time, an NAD+ molecule produced at the end of glycolysis gains a hydrogen atom to produce NADH. Pyruvate decarboxylase is an enzyme used to release CO2 from the pyruvate, and in the presence of alcohol dehydrogenase (an enzyme present in baker’s yeast), the pyruvate completes its conversion into ethanol. What you are left with is:
Pyruvate + NAD+ + ADP = Ethanol + NADH + C02 + ATP
The NADH produced can be used in the glycolysis cycle for the next glucose sugar. If you recall, ATP is the energy coenzyme that’s more powerful than ADP.
NOTE: During glycolysis, one glucose sugar is split into two pyruvates, doubling the output. Therefore, one glucose molecule produces two molecules of ethanol, two molecules of carbon dioxide and two ATP.
Homofermentation of LAB
The homofermentative process uses Lactic Acid Bacteria (LAB) to convert carbohydrates into lactate. It follows the same Embden-Meyerhof Pathway (EMP) as respiration and alcohol fermentation to produce its pyruvates.

Homofermentation is very similar to ethanol fermentation. Instead, the enzyme, lactate dehydrogenase is produced by LAB. It catalyses the pyruvate to produce lactate and ATP. Carbon dioxide is not produced in this process, but the lactic acid derived from lactate has many benefits which we’ll discuss later.
Again, as the glucose molecule is broken into two pyruvates during the EMP, 2 lactate and 2 ATP molecules are produced for each monosaccharide processed.
Fructose and other hexoses
Bread’s main starch is maltose, which is two glucose sugars bonded. This is seconded by fructose, which, alongside glucose makes the disaccharide sucrose. Any simple sugar can follow the pathway of glycolysis. Fructose will enter the pathway a little later on as fructose-6-p is the second stage of the process. There is evidence to suggest that fructose operates two pathways. This involves sucrose producing lactate, acetate, ethanol, CO2, and mannitol. Feel free to explore the linked article.
Heterofermentative LAB
Instead of EMP, Heterofermentative bacteria can use the Phosphoketolase Pathway (PKP). Sugars following this route produce:
- Lactate
- Carbon dioxide
- Either ethanol or acetate (leading to acetic acid).

In the PKP, glucose (a 6-carbon sugar) is reduced to a 5-carbon sugar (xylulose), releasing one carbon as CO2 in the process. This produces one 3-carbon molecule (glyceraldehyde-3-phosphate) and the 2-carbon, acetyl. Glyceraldehyde-3-phosphate, like before, will produce pyruvate which is converted into lactate and then, lactic acid.
Acetyl will produce acetic acid or follow the pathway further to make acetaldehydes (smelly substance) and then ethanol.
A process called co-metabolism is utilised for acetyl to transform into acetic acid. This is when a carbon backbone (a hexose sugar) and a co-substrate combine to facilitate the formation of acetic acid. If there is no suitable co-substrate, acetyl turns into ethanol. Possible co-substrates include:
- Fructose
- Pentoses
- Oxygen
- Citrate
- Malate
- Short-chain Aldehydes
- Oxidised Glutathione
But it doesn’t have to stop here. If the dough contains the correct Acetic Acid Bacteria, such as the genera known as acetobacter, ethanol can be oxidised to release carbon dioxide and water by following the Krebs cycle! And more gas means a faster rise!
Fermenting other sugars
Up till now, we’ve only considered the 6-carbon groups of monosaccharides categorised as hexoses. There are also a 5-carbon group of monosaccharides present in bread dough called pentoses. Blocks of pentoses are known as pentosans. These are readily found in rye flour and the bran of wheat flour. We’ll discuss the power of rye flour in sourdough fermentation near the end of this article.
Pentoses can steal a carbon to enter at the start of either glycolysis pathway. They are more likely, however, to break down into ribulose-5-phosphate to enter the PhosphoKetolase Pathway. The species and strain of the bacteria impact which sugars can be utilised and the preferred fermentation route.
Putting all fermentation pathways together!
Here is a diagram that provides the basic inputs and outputs of sugars in sourdough fermentation.

Starter or bread – What’s the difference?
These explain how wild yeasts and acid bacteria respire and ferment. This could be in a starter or when making sourdough bread.
The lifecycle of a new sourdough starter
A new starter starts with flour, water and lots of potential! See my sourdough starter recipe if you’ve not made your own starter before. Wild yeasts and LAB arrive in the starter through flour, water, air or transferred by equipment or hands. A new starter is mainly populated by wild yeasts during the first couple of days. After this, acid bacteria begin to multiply and overtake yeast activity. This leads to accelerating rates of fermentation, which produce ethanol and acids that lower the starter’s pH value.
As ethanol and acidity increase, the original strains of yeast and LAB struggle to operate in the newly hostile environment. Instead, strains more preferential to these conditions begin to thrive. There is a delay between conditions changing and the starter populating more robust strains, which accounts for the varied activity often noted during the first couple of weeks of a new starter.
A mature starter will be between 3.5 and 4.2 pH. Once the pH drops below 4.2 any harmful bacteria that may have multiplied during fermentation are killed off, and the starter is ready to use. Not seeing any life in your starter? Try my sourdough starter troubleshooting and fixes!
What happens when a starter peaks?
As the starter ferments, it produces CO2, which gets trapped in the gluten and forces the starter to rise. The starter will stop rising when it:
- Runs out of sugars to consume
- Acidity and ethanol levels rise too high, which inhibits the enzymes and fermentation processes
- Creates so much lactic acid that the gluten strands break down
When reaching the top of its rise, your starter will sit for an hour or two. At this point, little gas can be produced. In fact, the amount of gas created matches the amount that is lost from the structure, making it sit at the same height.
Leaving the starter at its peak before refreshing produces a more active and acidic tasting levain as acid bacteria continue to multiply and ferment. Eventually, lactic acids (and sometimes starving yeast) consume the gluten, and the starter collapses.
The impact of the regularity of refreshments in a starter
From the moment a starter is fed to the point that it peaks, the population of acid bacteria increases. This means that a starter that is regularly fed when at its peak will be more acidic, flavorful and powerful.
Common mistakes made by new sourdough bakers
Regular refreshments with the same flour make a starter more vibrant. This is due to the flour containing the microflora that the starter is used to consuming. Foreign materials are broken down by the starter, however, energy has to be used to create new enzymes to consume the foreign materials. This delays the starter’s rise and weakens its leavening power.
One mistake many bakers make is to change the flour used to feed their starter. This change in minerals means the starter has to alter the enzymes it produces thus weakening the starter until the perfect balance is achieved.
Using the same flour in your starter is best to make sourdough bread. It’s not the end of the world if you switch up your flours. Just expect a slower rising time. If you run into problems, see troubleshooting your sourdough bread. Another mistake is not keeping things clean. A dirty starter jar will introduce unwanted bacteria into the starter.
Types of yeast and lactic acid bacteria in sourdough
A sourdough starter usually contains between 1 to 4 different strains of yeast and lactic acid. There are hundreds of varieties available, with many believed still not discovered. The selection of strains found in your starter are based on your environment, the ingredients used and the conditions it is kept. I’ll discuss each of these in a moment, but for now, let’s consider what makes LAB strains different?
Lactic bacteria strains fall into one of three categories:
Obligate homofermentative
This type of LAB is largely found in new starters and not established ones. The strains ferment hexose sugars through the EMP pathway and almost entirely to lactic acid. Pentoses and gluconate cannot be fermented.
Obligate heterofermentative
These ferment hexose sugars almost entirely into lactic acid through heterofermentative routes. Under glucose-limiting conditions, they can produce CO2, lactic acid, acetic acid and ethanol. Pentoses can be fermented also.
Facultative heterofermentative
These are lactobacilli which ferment hexoses to produce lactic acid, CO2, acetic acid and ethanol through heterofermentation. They can also switch to following homofermentative routes to produce only lactic acid. Pentoses can be fermented to lactic acid and acetic acid. Processing routes can occur interchangeably, swapping from homofermentative to heterofermentative depending on the environment in your starter.
Common lactic acid bacteria in sourdough
- Lactobacillus fermentum
- Fructilactobacillus sanfranciscensis (previously known as “Lactobacillus sanfranciscensis”)
- Pediococcus pentosaceus
- Lactobacillus plantarum
- Lactobacillus brevis
The yeast strains found in a culture vary between starters too. These also generate flavours and aromas that are unique to each. Here are some of the most popular yeast strains found in sourdough starters:
Common yeast strains in sourdough
- Kazachstania exigua (Saccharomyces exiguous)
- Saccharomyces cerevisiae (commercial yeast)
- Candida milleri
- Candida humilis
The impact of different bacteria and yeasts
In an established culture, the yeasts and LAB will complement each other. For example, the most widespread lactobacilli (LAB), lactobacillus sanfranciscensis (now called “Fructilactobacillus sanfranciscensis”) separates maltose into a glucose-1-phosphate and a glucose molecule. The glucose-1-phosphate portion enters the heterofermentative pathway to become glucose-6-phosphate, whilst glucose is utilised by the yeast. If fructose is available, acetic acid is produced from acetate. As fructose supplies deplete, the acetate will produce ethanol instead.
How fermentation benefits sourdough bread
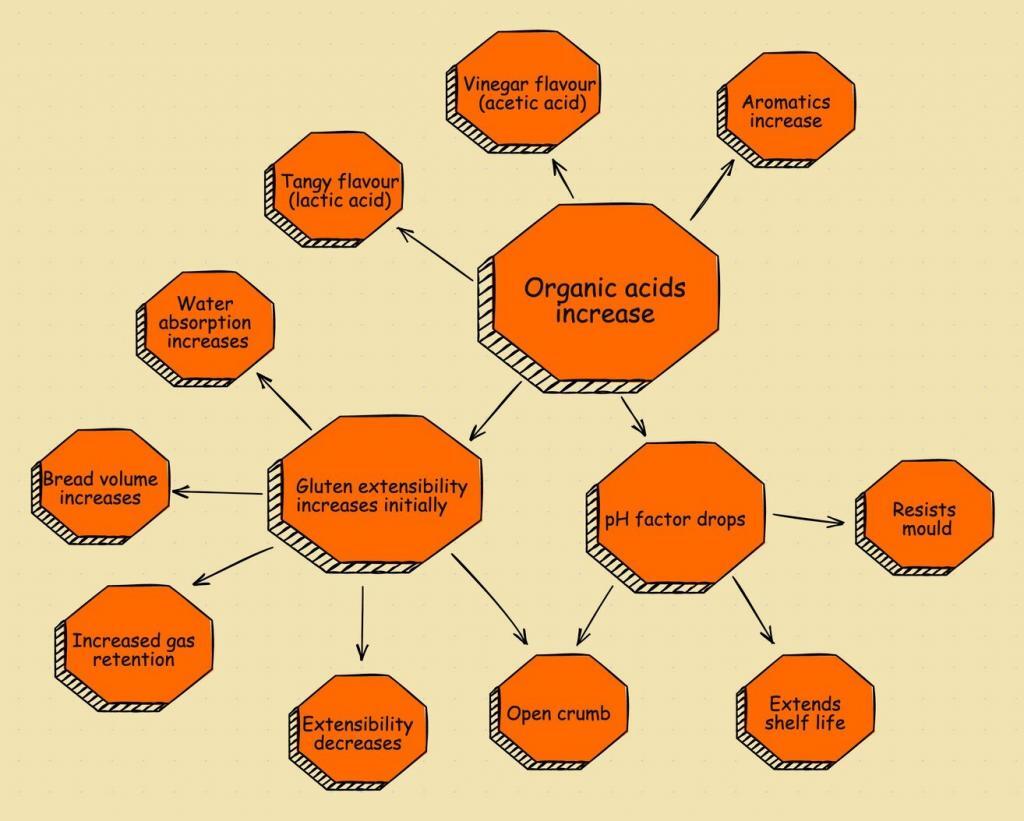
The basic necessity of sourdough bread production is simple. It uses a starter capable of producing CO2 to make the dough rise. But there’s more to a sourdough starter than simply rising the dough. What about the health benefits of sourdough? The unique tangy flavour? Or the texture of sourdough bread? Now we have understood the science behind what makes a starter a levain, let’s cover the key components that make it so special.
Lactic acid
Lactic acids are vitally important for the dough as they provide maturation. Based on an experiment by Bakerpedia, lactic acid provides the following benefits to sourdough:
- Increases acidity
- Enhances flavour
- Adds flavour
- Prevents mould, which increases shelf-life
- Produces dough conditioners
- Helps to absorb minerals
- Retains gas
Acetic acid
Maybe you are familiar with the similarity between acetic acid and vinegar? Well, vinegar is (4%) acetic acid that’s diluted with water. Its production in sourdough fermentation produces CO2, but it also benefits the quality of the bread by:
- Lowering the pH to kill harmful bacteria
- Improving shelf life
- Producing a twangy aroma
- Adding sour flavour notes
You can see more about the effects of adding vinegar to dough here.
Ethanol (alcohol)
Ethanol is utilised in the maturation of the dough. It is not (as many claim), a by-product of the fermentation process. It is absolutely needed! After the bread is baked, only 2% of the ethanol remains. It is used to:
- Improve odour
- Add flavour
- Control unwanted bacteria, which improves keeping quality
ATP
The Adenosine TriphosPhate produced during fermentation is utilised at the start of each cycle of reactions. It provides energy by donating a phosphate molecule so glycolysis processes can repeat.
Cereal enzymes
As the environment becomes more acidic, cereal enzymes become more active. Here are a few of the important ones:
Phytase
Phytase hydrolyses fructose, ATP and phytic acid. Flour contains between 1-1.5% of
phytic acid, which binds dietary minerals such as iron, calcium, and zinc together. Hydrolising phytic acid with phytase breaks the bonds to aid mineral absorption when sourdough bread is eaten.
Gluconic acid
Gluconic acid, formed by the acidification of glucose is used in cement! It strengthens the dough and enhances the gas retention properties of the gluten.
Proteases
If you use autolyse in sourdough, you may already understand the impact of proteases. These protein enzymes catalyse the breaking down of peptide bonds between gluten strands through hydrolysis. This speeds up the rate proteins are broken down into polypeptides or single amino acids.
Smaller protein chains make the dough more stretchy (extensible), and any loose amino acids make the dough feel softer. In autolyse, this, alongside the hydration of the starch and protein shortens the kneading time. Free amino acids can be processed by the yeast to improve crust colour and flavour through Malliard reactions.
Prentosoraces
Pentosans are carbohydrates found predominantly in rye flour, consisting of 20-30% of the grain. They are also found in the bran of whole wheat flour. They initially store a lot of water, diverting from the flour’s protein.
Pentosans play a central role in creating the structure of bread in the oven before it passes 45C (113F). Prentosorace enzymes multiply as the dough warms and break down pentosans, releasing the stored water. This is why rye bread dough becomes stickier as it ferments and (partially) why it is suspect to collapse in the oven.
How to change a sourdough starter’s characteristics
Flour
The ash levels of flour are measured by burning the flour and measuring what is left. The ash that remains is the minerals it contains. Minerals have t be digested by the yeast and bacteria which slows the production of fermentative products. Slowing the rate of fermentation increases the number of organic acids produced, making a more active starter. Less speed, more gas!
Ash is found in higher quantities in whole wheat, rye and high protein flours. As rye is high-ash, many bakers switch 10-30% of the white flour for rye flour in their starters.
Malted barley/malt flour
Malted barley flour is sometimes added to sourdough bread recipes. When malting barley, you produce a powerful source of ɑ-amylase which is then dried and sold as malt flour. It can be used to support flour that has reduced enzyme activity.
I must highlight that I often see sourdough bakers attempting to follow recipes that contain activated malt flour. The purpose of this ingredient in sourdough bread fermentation is to enhance a particular flour. For most situations, the best solution is to select high-quality flour and add malt flour if required. The overproduction of maltose (that it creates) leads to a gummy or tacky crumb that’s most unpleasant.
Salt
Salt impacts the strains of yeast and bacteria in a sourdough starter or dough. By diverting water from the yeast and bacteria cells, salt creates osmotic pressure. Some strains will prefer the environment more than others which changes the levains characteristics. Some starter recipes include salt, there is no argument to include it, but expect a different collection of bacteria and yeasts to evolve.
By slowing down the rate of transfer between the yeast and the water cells (or bacteria and water), the rate of fermentation is also slowed.
Aside from fermentation changes, salt is one of the basic senses so is a flavour enhancer. It also helps to keep products fresh.
The gluten structure is also tightened with the addition of salt. This strengthening allows the dough to contain carbon dioxide better and improves loaf volume during oven spring.
Water
The amount of mineral activity and acidity in water affects the rate of sourdough fermentation. Higher levels of minerals found in hard water areas produce bread with a slower, yet higher rise and more flavour. The format is similar to the popularity of beer brewers in Buxton, UK.
Scientifically this happens, but finding a significant deterioration or advantage is rare for home bakers. Providing the water is drinkable and not overly chlorinated it is perfect for making sourdough. I don’t filter mine! If you suspect that your tap water is bleached or chlorinated at high levels, leave it out for 30 minutes before using it, or switch to bottled water.
The impact of the length of fermentation in sourdough bread
The level of fermentation increases with the length of time dough ferments. Increasing the length of fermentation allows the sourdough to develop more flavour and gas retaining features. Too much fermentation can result in the gluten breaking down, and the dough collapsing.
How temperature changes sourdough
Bar changing the amount of starter used, temperature is the most common way to tweak the rate at which sourdough ferments. Warming up sourdough increases the rate of fermentation and also, the rate at which the enzymes break down the sugars so the yeast and bacteria can be “fed”.
We can cool the temperature of the dough to slow down fermentation. This method can be used to overnight “retard” sourdough bread to be baked the following day, or by weekend bakers who store their starter in the fridge so they don’t have to feed it as often. Yet timing isn’t the only reason that the temperature of sourdough should be considered. Temperature changes the flavour of sourdough too.
Be it, obligate homofermentative, obligate heterofermentative or facultative heterofermentative bacteria types, they all prefer to be warmth. A more viscous dough also allows the bacteria to operate at a faster rate too.
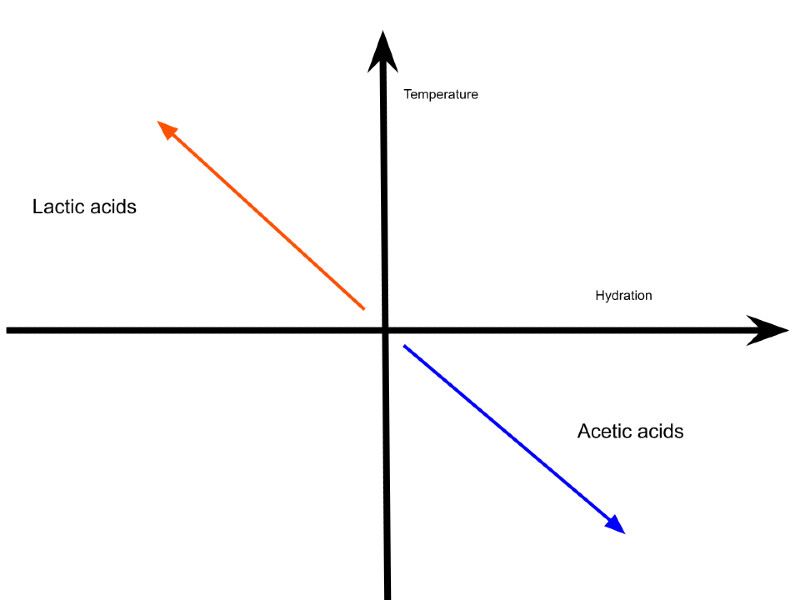
To alter the flavour of sourdough bread we can lower the activity of certain fermentation bacteria, allowing others to become more prevalent. A dryer environment means the transfer of cells is slowed down. This slows all three fermentative bacteria types. As homofermentative bacteria only produce lactic acid, less viscosity leads to slower lactate production, allowing acetic acid production to increase in heterofermentative reactions.
Here’s a table that explains what occurs when water and temperature are increased or decreased together:
| Fermentative bacteria | Result when water and temperature are increased | Result when water and temperature are decreased |
| 0. Homofermentive | More lactic acid is produced | Lactic acid production slows |
| 0. Heterofermentive | Lactic acid and alcohol production increases | Acetic acid production increases |
| F. Heterofermentative | Only lactic acid is produced | Only acetic acid is produced |
| Combined results: | More lactic acid | More acetic acid |
Both heterofermentative bacteria types produce more acetic acid in a starter/dough when water and temperature are decreased. But really it’s a case of a lack of water slowing all acid production routes down and cooler temperatures boasting acetic acid production.
To be clear, a decrease in temperature and viscosity doesn’t boast acetic acid production. The same fermentative bacteria will operate faster at a warmer temperature! The reason for the increase in acetic acid is based on the supply of sugars available for fermentation.
In addition to altering the fermentative bacteria to produce more acetic acid in your starter cooler temperatures also play a factor by converting more acetyl into acetic acid instead of ethanol. Acetyl needs to be co-metabolised, which is usually done by fructose, primarily produced by enzymes supplied by the yeast. The enzymes break sucrose into glucose and fructose molecules. The enzymes (mainly xylose), continue to operate when the dough is cool, albeit at a slower rate. This means more fructose is available for producing acetic acid. As the dough warms up, fructose can be fermented by yeast or bacteria, those that are consumed will leave a sweeter flavour in the bread.
Wet bread dough proofed at warmer temperatures rises faster and generates more lactic acid – a creamy-sharp yoghurt-like flavour.
Dough that is dryer and fermented cooler will have a vinegary and acidic flavour.
Fermenting sourdough in the fridge
Reducing the temperature of sourdough below 3C (37F) halts enzymic activity. Placing sourdough in the refrigerator overnight helps with timing as you’ll be able to bake your bread for lunch or breakfast without having to work through the night. Whilst enzymes are temporarily inactive, hydrolysis continues to simplify starch into smaller sugars. Once the dough warms the sugars can be utilised by the yeast and LAB, providing a short boost in activity. Any excess sugars will provide a sweeter flavour, or be utilised with proteins for extra browning when the bread is baked in the oven. Slowing down leavening activity provides gluten time to improve.
Placing a ¾ proofed sourdough bread in the fridge overnight will slow down gas production. Yet as there will be plenty of lactate already produced, they will continue to multiply and make the dough more acidic. Find out how more on how to regulate the temperature in the desired dough temperature and bread proofing temperature articles.
Over proofing sourdough
There are many benefits of extending sourdough fermentation. But alongside the ups, there are also downsides and the risk of over-proofing sourdough.
As fermentation continues, the dough will absorb more oxygen. This provides an initial boost in gluten elasticity. But as time goes on it destroys the carotenoid pigments and weakens the dough.
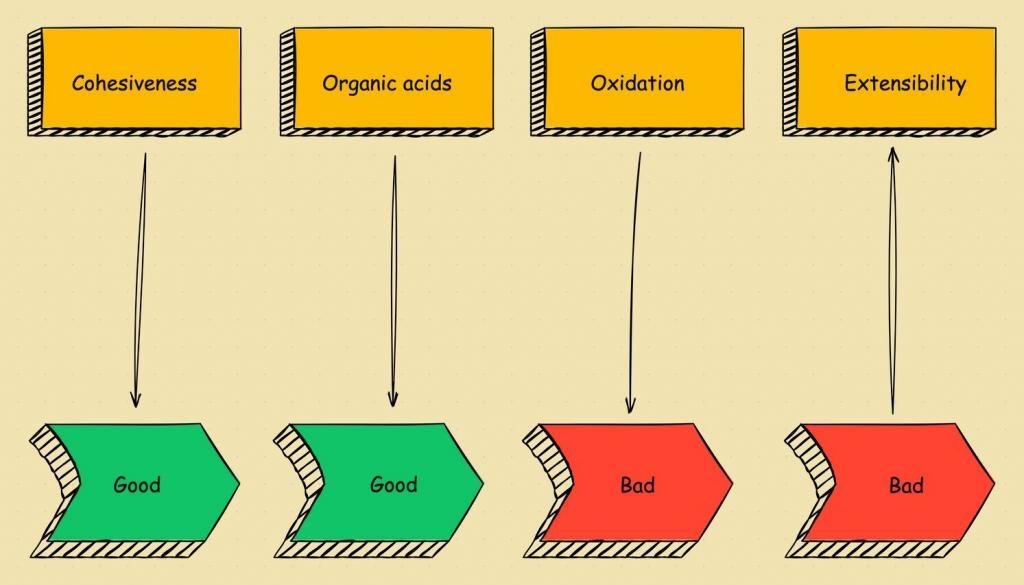
When sourdough becomes over-proofed the yeast and bacteria exert all available sugars. “This means that the bread will stop rising, right?” Well, no, not really! Instead of breaking down starch, the levains start to consume gluten proteins. This weakens the structure of the gluten and the result at best is big bubbles in the bread but often leads to the dough structure collapsing.
Sourdough baking timetables

These are typical baking schedules for sourdough baking. Note that the fermentation time is increased or lowered when the dough is kneaded more or less. A proofing temperature in the region of 28C (82F) is used in these examples except for the heavy knead method where a proofer set at 35C (95F) is used.
No-knead – fridge final proof
- Autolyse 30 mins
- Kneading/incorporation 2 mins
- Bulk fermentation 4 hours
- Shaping 30 mins
- Fridge final proof 8 hours
- Bake 35 mins
Total 13 hours 37 minutes
No-knead – fridge
- Autolyse 30 mins
- Kneading/incorporation 2 mins
- Bulk fermentation 3 hours
- Fridge fermentation 6 hours
- Shaping 40 mins
- Final proof 4 hours
- Bake 35 mins
Total: 14 hours 47 minutes
Lightly knead
- Autolyse 30 mins
- Kneading 6 mins
- Bulk fermentation 4 hours
- Shaping 30 mins
- Final proof 5 hours
- Bake 35 mins
Total: 10 hours 41 minutes
Heavily knead
- Autolyse 15 mins
- Kneading 10 mins
- Bulk fermentation 2 hours
- Shaping 30 mins
- Final proof 3 hours
- Bake 35 mins
Total: 6 hours 30 minutes
Knead & in fridge
- Autolyse 20 mins
- Kneading 6 mins
- Fridge Bulk fermentation 12 hours
- Bulk rise 2 hours 30 mins
- Shaping 30 mins
- Final proof 4 hours
- Bake 35 mins
Total: 20 hours 01 minutes
Conclusion: Is sourdough bread better than yeast bread?
There are fewer gas-producing components in sourdough than in yeast bread, which slows its rising ability. Because of this extended production time, a more flavoursome and healthier product is produced. Sourdough bread typically has a lower amount of gluten (low gi), less starch and keeps fresher for longer than yeast bread. It is also less likely to contain further fortification through artificial dough conditioners as it conditions the dough itself. So yes, I think sourdough is better than yeast bread, what do you think? Let me know in the comments below:
Additional references:
The Taste of Bread – Raymon Calvel
https://www.thefreshloaf.com/node/10375/lactic-acid-fermentation-sourdough
Sourdough Fermentation – Bakerpedia
Sourdough fermentation process frequently asked questions
If you’ve enjoyed this article and wish to treat me to a coffee, you can by following the link below – Thanks x

Hi, I’m Gareth Busby, a baking coach, head baker and bread-baking fanatic! My aim is to use science, techniques and 15 years of baking experience to help you become a better baker.
Table of Contents
- What is sourdough?
- What is in a sourdough starter?
- How does sourdough produce gas?
- Stage 1: Breaking down starch into sugars
- Step 2: Glycolysis breaks down sugars for respiration and fermentation
- Step 3: Fermentation processes
- Putting all fermentation pathways together!
- The lifecycle of a new sourdough starter
- What happens when a starter peaks?
- Common mistakes made by new sourdough bakers
- Types of yeast and lactic acid bacteria in sourdough
- The impact of different bacteria and yeasts
- How fermentation benefits sourdough bread
- How to change a sourdough starter’s characteristics
- Over proofing sourdough
- Sourdough baking timetables
- Conclusion: Is sourdough bread better than yeast bread?
- Sourdough fermentation process frequently asked questions
Related Recipes
Related Articles
Keep up to date with the latest Articles, Recipes & Bread Baking info by joining my mailing list
Join The Weekly Bread Baker's Newsletter!
Latest Articles
Baking Categories
Disclaimer
Address
8 Woodland Avenue,
Worthing
West Sussex
BN13 3AF
UK







